
- What is an PEM electrolyser, and how does it work?
- How does an PEM electrolyser differ from alkaline and AEM technologies?
- What are the key components of a PEM electrolyser stack?
- What materials are used in PEM electrolyzers and why are precious metals required?
- What are the typical operating conditions of PEM electrolyzers?
- How is performance and efficiency evaluated in PEM electrolyzers?
- What are the main degradation mechanisms in PEM electrolyzers?
- What safety considerations are important in PEM electrolyzers?
- What is the role of the balance of plant (BoP) in PEM systems?
- What are the cost drivers and commercialization challenges?
- Where are PEM electrolyzers best applied?
- What is the future outlook for PEM electrolyzer technology?
What is an PEM electrolyser, and how does it work?
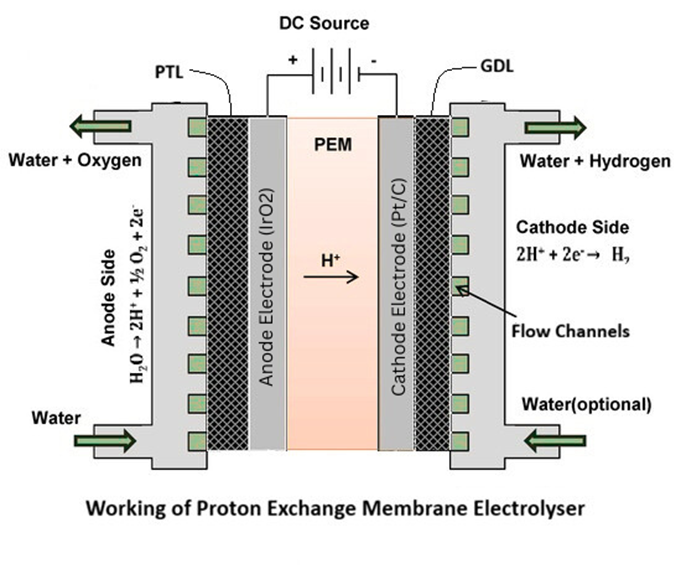
A Proton Exchange Membrane (PEM) electrolyzer is a water electrolysis technology that produces hydrogen by splitting water molecules using direct current (DC) electricity. It operates using a solid polymer membrane that conducts protons (H⁺) while separating hydrogen and oxygen gases. When DC power is applied, water is supplied to the anode side of the cell. At the anode, water undergoes oxidation to produce oxygen gas, protons, and electrons. The reaction at the anode is:
2H₂O → O₂ + 4H⁺ + 4e⁻
The protons generated at the anode migrate through the proton-conducting membrane to the cathode. The membrane allows only protons to pass while blocking gases and electrons. The electrons travel through the external circuit and reach the cathode, where they combine with protons to produce hydrogen gas:
4H⁺ + 4e⁻ → 2H₂
The membrane serves three critical functions: proton conduction, gas separation, and electrical insulation between electrodes. Because the electrolyte is solid and integrated into the membrane, PEM electrolyzers have a compact structure, fast response time, and high operational efficiency. The acidic environment inside the cell requires highly corrosion-resistant materials, which strongly influences system design.
How does an PEM electrolyser differ from alkaline and AEM technologies?
PEM electrolysis differs significantly from alkaline and Anion Exchange Membrane (AEM) electrolysis in ion transport mechanism, operating chemistry, materials, and system behavior. PEM systems conduct protons (H⁺) through a solid membrane in an acidic environment. In contrast, alkaline systems conduct hydroxide ions (OH⁻) through a liquid potassium hydroxide electrolyte. AEM systems also conduct hydroxide ions but use a solid membrane instead of liquid electrolyte.
PEM electrolyzers are compact and capable of operating at high current densities. They respond rapidly to power fluctuations, making them highly suitable for renewable energy integration. However, the acidic conditions require expensive platinum-group metal catalysts such as iridium and platinum, as well as titanium hardware for corrosion resistance.
Alkaline systems are less expensive in terms of materials but are bulkier and slower to respond dynamically. AEM systems aim to combine the cost advantages of alkaline systems with the compact structure of PEM systems but are still under development.
Thus, PEM technology is considered the most mature and performance-oriented option for high-pressure and dynamic hydrogen production.
What are the key components of a PEM electrolyser stack?
A PEM electrolyzer stack consists of multiple repeating electrochemical cells connected electrically in series. Each cell contains several critical components. At the center is the proton exchange membrane, typically made from a perfluorosulfonic acid polymer such as Nafion. This membrane conducts protons while separating hydrogen and oxygen gases.
On either side of the membrane are catalyst layers. The anode catalyst facilitates the oxygen evolution reaction, while the cathode catalyst supports the hydrogen evolution reaction. These catalyst layers are supported by porous transport layers (PTLs) or gas diffusion layers (GDLs) that distribute water and remove gases efficiently.
Bipolar plates are positioned between adjacent cells. They distribute current uniformly, manage fluid flow through internal channels, and provide structural support. In PEM systems, bipolar plates are often made of titanium due to the corrosive acidic environment.
Seals and gaskets prevent gas leakage and maintain separation between compartments. The stack is compressed using end plates and tie rods to ensure uniform contact pressure. Proper compression is critical for electrical performance, sealing, and long-term durability.
What materials are used in PEM electrolyzers and why are precious metals required?
The acidic environment in PEM electrolyzers requires highly stable and corrosion-resistant materials. The membrane is typically made from fluorinated polymers such as perfluorosulfonic acid membranes. These materials offer high proton conductivity, chemical stability, and mechanical durability.
At the anode, iridium oxide is commonly used as the catalyst because it can withstand the harsh oxidative acidic conditions of the oxygen evolution reaction. At the cathode, platinum is typically used due to its excellent hydrogen evolution activity and stability.
Titanium is widely used for bipolar plates and porous transport layers on the anode side because it resists corrosion in acidic and oxidative environments. Stainless steel cannot be used directly at the anode because it would corrode rapidly.
The need for platinum-group metals significantly increases system cost. Catalyst loading and iridium scarcity are major research concerns in PEM technology.
What are the typical operating conditions of PEM electrolyzers?
PEM electrolyzers typically operate at temperatures between 50°C and 80°C. Higher temperatures improve reaction kinetics and proton conductivity but may accelerate membrane degradation. Operating pressures can range from atmospheric pressure to 30–70 bar, with some advanced systems operating even higher.
Current densities typically range from 1 to 3 A/cm², which is higher than alkaline systems. High current density enables compact stack design and higher hydrogen production per unit area.
PEM systems require high-purity deionized water. Impurities can poison catalysts and degrade the membrane. Cooling systems are integrated to remove heat generated during operation. Precise pressure control is essential to prevent gas crossover and maintain safety.
How is performance and efficiency evaluated in PEM electrolyzers?
Performance is mainly evaluated through cell voltage, hydrogen production rate, and energy efficiency. At a given current density, lower cell voltage indicates higher efficiency. The theoretical minimum voltage for water electrolysis is about 1.23 V, but practical PEM systems operate between 1.8 and 2.2 V depending on current density.
Efficiency losses include activation losses from reaction kinetics, ohmic losses from membrane resistance, and mass transport losses from gas and water flow limitations. Faradaic efficiency measures how effectively electrical current is converted into hydrogen.
Energy consumption typically ranges from 50 to 60 kWh per kilogram of hydrogen. Stack efficiency differs from system efficiency, as balance-of-plant components also consume energy.
What are the main degradation mechanisms in PEM electrolyzers?
Degradation in PEM electrolyzers mainly occurs in the membrane and the catalyst layers. These components operate under harsh chemical and electrochemical conditions. Over time, their performance slowly decreases.
Membrane degradation can occur due to chemical attack. Reactive radicals formed during operation can damage the polymer structure of the membrane. This reduces its proton conductivity. Mechanical stress is another cause of degradation. The membrane expands and contracts due to changes in temperature, hydration level, and pressure. Repeated stress can lead to thinning or small cracks. If the membrane becomes too thin or damaged, gas crossover may increase. This reduces efficiency and creates safety concerns.

Catalyst degradation is also a major issue. At the anode, iridium can slowly dissolve under high potential conditions. This reduces catalytic activity over time. Catalyst particles can also agglomerate, meaning they grow larger and lose surface area. In some cases, catalyst particles may detach from their support structure. This decreases the number of active reaction sites and increases cell voltage.
Mechanical stresses from pressure cycling and thermal expansion also affect stack durability. Frequent load changes can increase stress on both the membrane and electrodes. Over long periods, these effects reduce system performance. Under well-controlled operating conditions, PEM electrolyzer stacks typically achieve lifetimes between 50,000 and 80,000 operating hours. Actual lifetime depends on operating temperature, pressure, current density, and maintenance practices.
What safety considerations are important in PEM electrolyzers?
Safety in PEM electrolyzers mainly focuses on preventing hydrogen and oxygen from mixing. A mixture of hydrogen and oxygen can be explosive. The system must also control pressure carefully during operation. If pressure is not balanced, gases can cross through the membrane. This is called gas crossover. Gas crossover increases the risk of forming a dangerous mixture inside the cell.
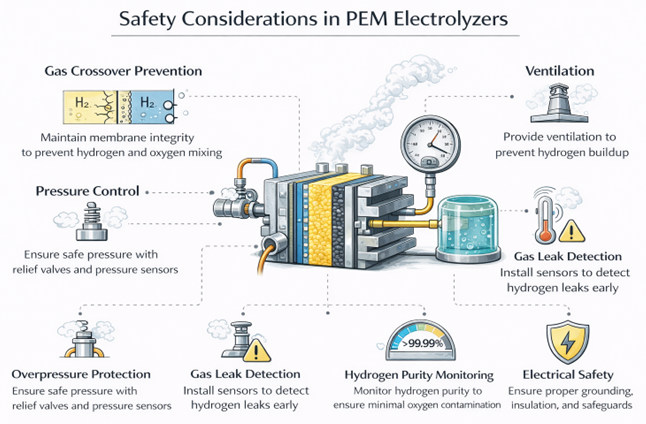
To prevent this, PEM systems use differential pressure control. This keeps the pressure difference between the anode and cathode within safe limits. Hydrogen purity is continuously monitored during operation. If oxygen is detected in the hydrogen stream, the system can trigger alarms or shutdown procedures. Leak detection sensors are installed around the system to detect any hydrogen leakage early.
Emergency venting systems are also included. These systems safely release gases if abnormal pressure builds up. Proper startup and shutdown procedures are very important. Sudden changes in pressure or temperature can damage the membrane and increase crossover risk. Controlled ramp-up and ramp-down reduce mechanical and thermal stress on the stack.
Many PEM electrolyzers operate at high pressure to reduce downstream compression needs. High-pressure operation requires strong mechanical design and high-quality materials. All pressure vessels, seals, and fittings must meet strict safety standards. Hydrogen is highly flammable and ignites easily. Therefore, careful safety engineering, monitoring, and maintenance are mandatory in PEM electrolyzer systems.
What is the role of the balance of plant (BoP) in PEM systems?
The balance of plant, or BoP, includes all the supporting systems required for proper operation of a PEM electrolyzer. These systems do not produce hydrogen directly, but they enable the stack to work safely and efficiently.
Power electronics are a key part of the BoP. Rectifiers convert alternating current (AC) from the grid or renewable sources into direct current (DC). The electrolyzer stack requires stable DC power for controlled operation. Power control systems also help manage startup, shutdown, and load changes. Water purification systems supply high-purity deionized water to the stack. Impurities in water can damage the membrane and poison the catalysts. Proper water treatment improves performance and extends system life.
Cooling systems remove the heat generated during electrolysis. Temperature must be kept within a specific range to protect the membrane and maintain efficiency. Heat exchangers and cooling loops are commonly used for this purpose. Gas–liquid separators remove water droplets from the hydrogen and oxygen streams after production. Dryers are used to reduce moisture content in the gas before storage or further use. In some systems, compressors are integrated to increase hydrogen pressure for storage or transport.
Instrumentation and control systems continuously monitor key parameters such as voltage, current, temperature, pressure, and gas purity. These systems allow automatic operation and quick detection of faults. The BoP can represent a large portion of the total system cost. It strongly affects overall efficiency, safety, and long-term reliability of the PEM electrolyzer system
What are the cost drivers and commercialization challenges?
The main cost drivers in PEM electrolyzers are precious metal catalysts, titanium components, and membrane materials. The anode catalyst usually contains iridium, and the cathode contains platinum. These metals are expensive and limited in supply. The amount of catalyst used in each cell, known as catalyst loading, has a strong impact on the total capital cost. Higher catalyst loading increases system cost directly.
Titanium is widely used in PEM systems, especially for bipolar plates and porous transport layers on the anode side. Titanium is corrosion-resistant in acidic conditions, but it is also costly. This adds to the overall stack cost. The proton exchange membrane itself is made from specialized fluorinated polymers, which are also expensive to manufacture.
Iridium scarcity is a major concern for large-scale deployment. Global iridium production is limited, and increasing demand from PEM electrolyzers may create supply chain risks. This makes catalyst reduction and alternative materials a major research focus.
To reduce cost, large-scale manufacturing and automation are necessary. Mass production helps standardize components and reduce labour cost. Higher production volumes also reduce material cost through bulk purchasing. As manufacturing scales up, stack cost can decrease due to economies of scale. This is essential for making PEM hydrogen production economically competitive
Where are PEM electrolyzers best applied?
PEM electrolyzers are well suited for applications that require very high purity hydrogen. The hydrogen produced is typically of high quality and can be used directly in fuel cells and other sensitive applications. These systems also have a compact design, which makes them suitable for installations where space is limited.
PEM electrolyzers respond quickly to changes in power input. They can increase or decrease production within seconds. This makes them ideal for dynamic operation. They are widely used in green hydrogen projects that are connected to wind and solar power plants. Renewable energy sources often produce fluctuating power, and PEM systems can handle these variations effectively.
PEM electrolyzers are also suitable for high-pressure hydrogen production. They can produce hydrogen at elevated pressures, which reduces the need for additional compression. This is useful in hydrogen mobility applications such as fuel cell vehicles and refuelling stations.In addition, PEM systems are used for grid balancing services. They can absorb excess electricity when renewable generation is high and convert it into hydrogen. Their fast response time and operational flexibility make them highly compatible with modern renewable energy systems.
What is the future outlook for PEM electrolyzer technology?
The future of PEM electrolysis depends on several important improvements. One major goal is to reduce catalyst loading. Precious metals such as iridium and platinum are expensive and limited in supply. Lowering the amount of these metals in each cell will significantly reduce system cost. Researchers are also working on developing alternative catalyst materials that use less or no iridium.
Improving membrane durability is another key priority. The membrane must operate for many years without losing conductivity or mechanical strength. Better chemical stability and resistance to mechanical stress will help extend stack lifetime. Longer lifetime reduces replacement cost and improves overall project economics.
Manufacturing cost reduction is equally important. Large-scale automated production can lower unit cost through standardization and economies of scale. Many companies are investing in gigawatt-scale manufacturing facilities to increase production capacity. High-pressure operation is also being further developed. Producing hydrogen directly at higher pressure reduces the need for external compressors. Modular stack designs are becoming more common. These designs allow easier scaling and maintenance.

 Login
Login












